Roeffaers Lab
Nanoscopy and catalysis
Publication
Article Published in ACS Energy Letters
Abstract
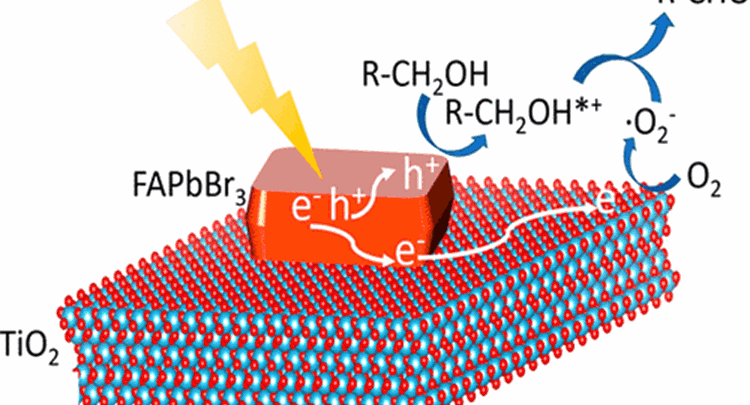
The impressive optoelectronic performance and low production cost of metal halide perovskites have inspired applications well beyond efficient solar cells. Herein, we widen the materials engineering options available for the efficient and selective photocatalytic oxidation of benzylic alcohols, an industrially significant reaction, using formamidinium lead bromide (FAPbBr3) and other perovskite-based materials. The best performance was obtained using a FAPbBr3/TiO2 hybrid photocatalyst under simulated solar illumination. Detailed optical studies reveal the synergetic photophysical pathways arising in FAPbBr3/TiO2 composites. An experimentally supported model rationalizing the large conversion enhancement over the pure constituents shows that this strategy offers new prospects for metal halide perovskites in photocatalytic applications.
DOI: 10.1021/acsenergylett.8b00131